Adiabatic Heating and Cooling
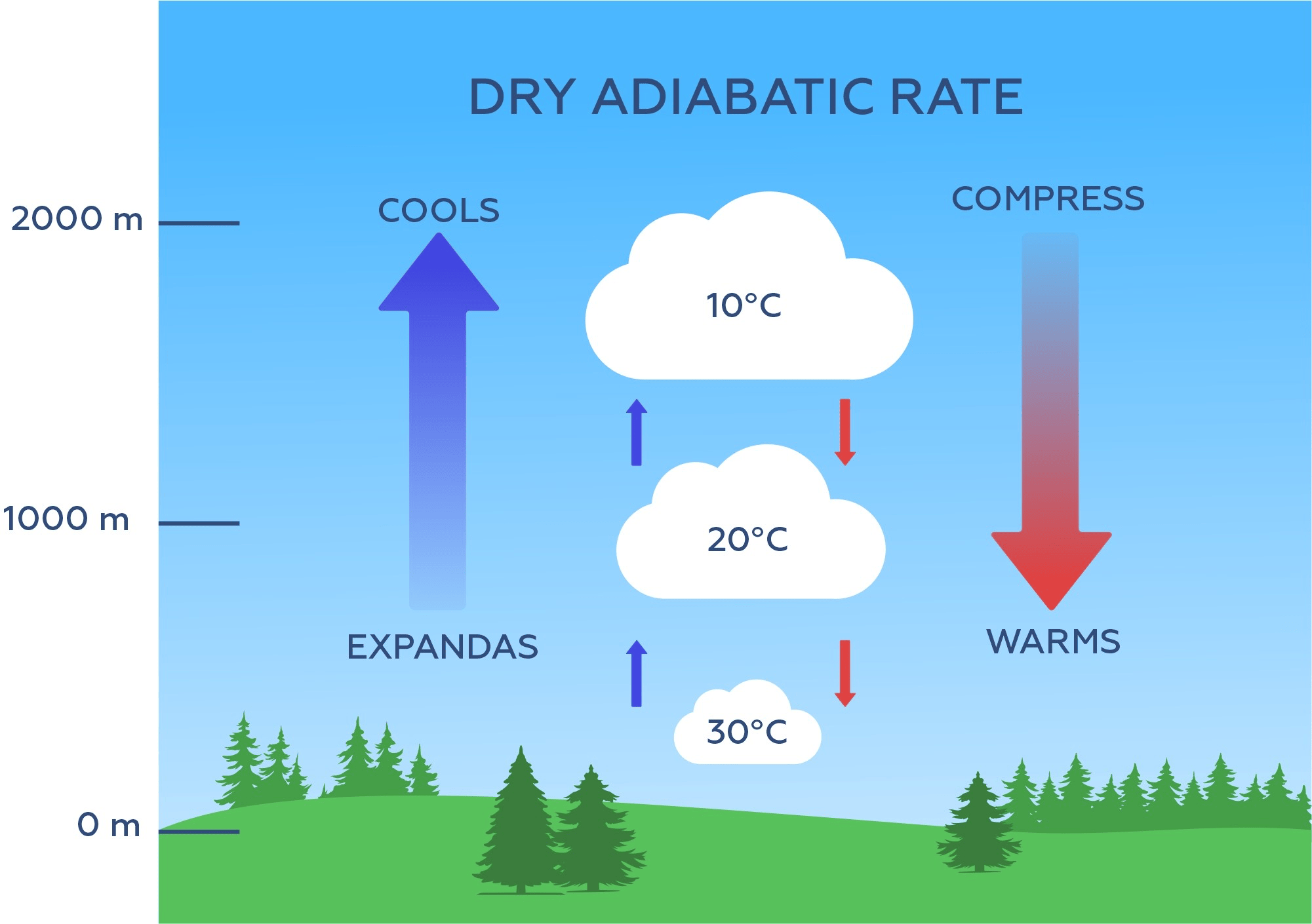
To begin, let’s start with a diagram from Epic’s online course. In this diagram, you see an orange parcel of air down near the surface at 30°C. As it rises through the air, it cools. Conversely, if you start at the top of the diagram, you see a large blue parcel of air at 10°C, and as it descends through the air, it warms.
This is a result of what we call adiabatic heating and cooling. Adiabatic heating or cooling occurs when a volume of air expands as it rises or compresses as it descends, without exchanging any heat with the surrounding atmosphere.
Atmospheric Stability
The following diagram is referred to Aviation Weather Handbook (FAA-H-8083-28A)
Absolute Stability
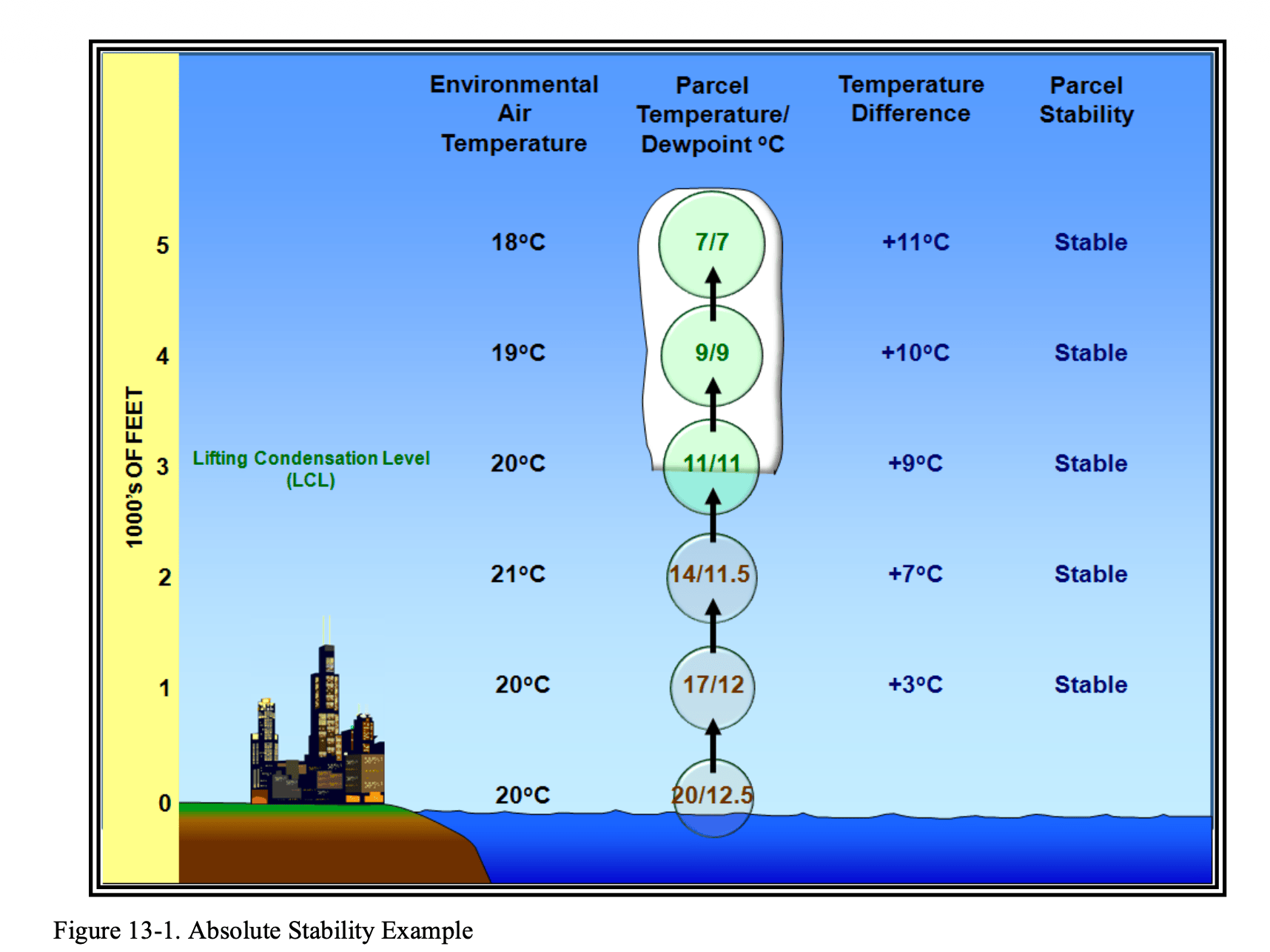
In an example of absolute stability, imagine an air parcel at the surface with a temperature of 20°C and a dew point of 12.5°C. As that air parcel rises, the temperature and dew point decrease until they reach 11°C at the base of a cloud. If we compare the temperature of the cooling parcel to the surrounding atmospheric air, and the difference is positive (the parcel is cooler than the environment), the air is stable.
Neutral Stability
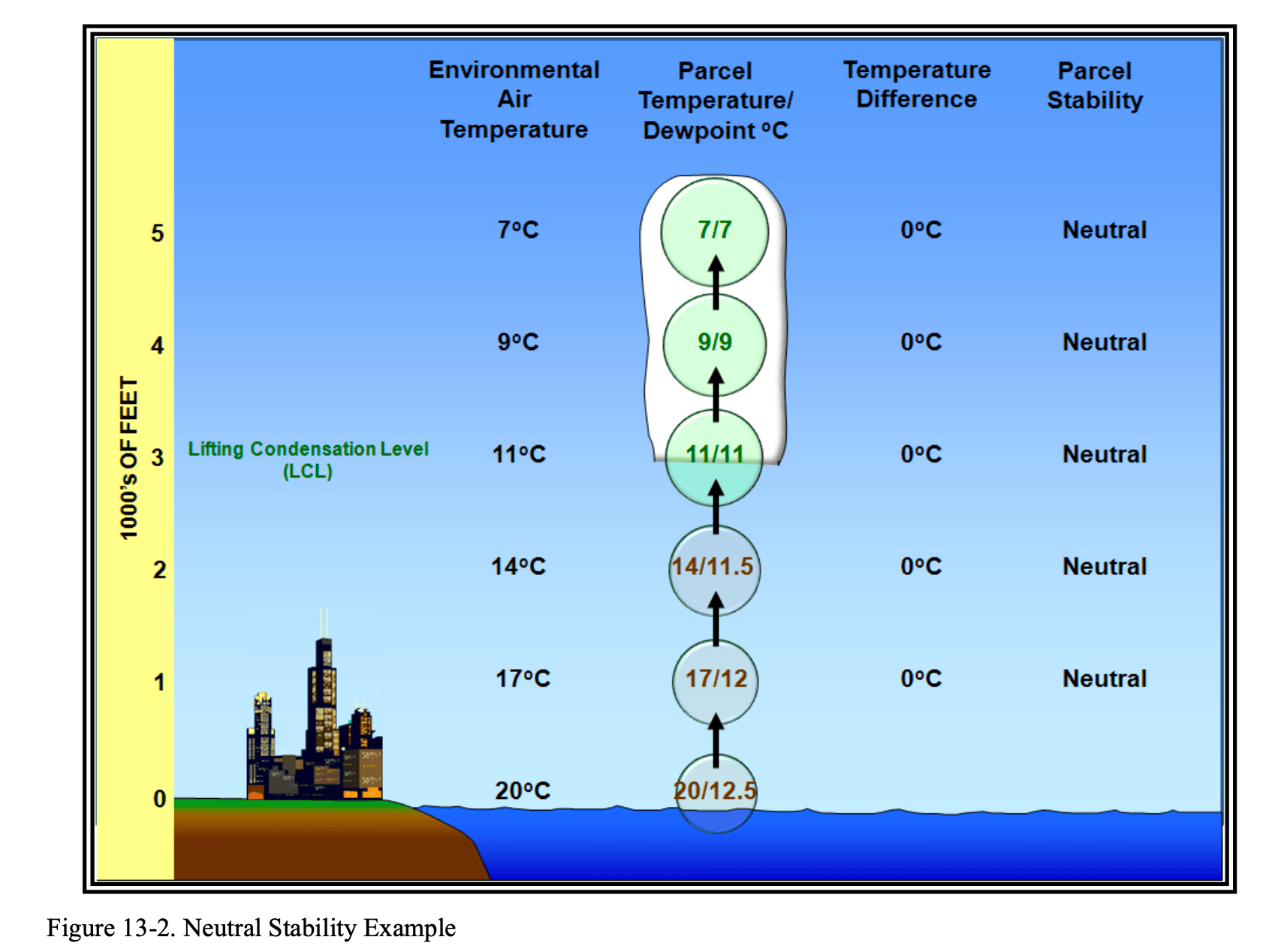
In cases of neutral stability, as the parcel rises and cools adiabatically, the difference between the environmental air temperature and the parcel temperature is zero.
Absolute Instability
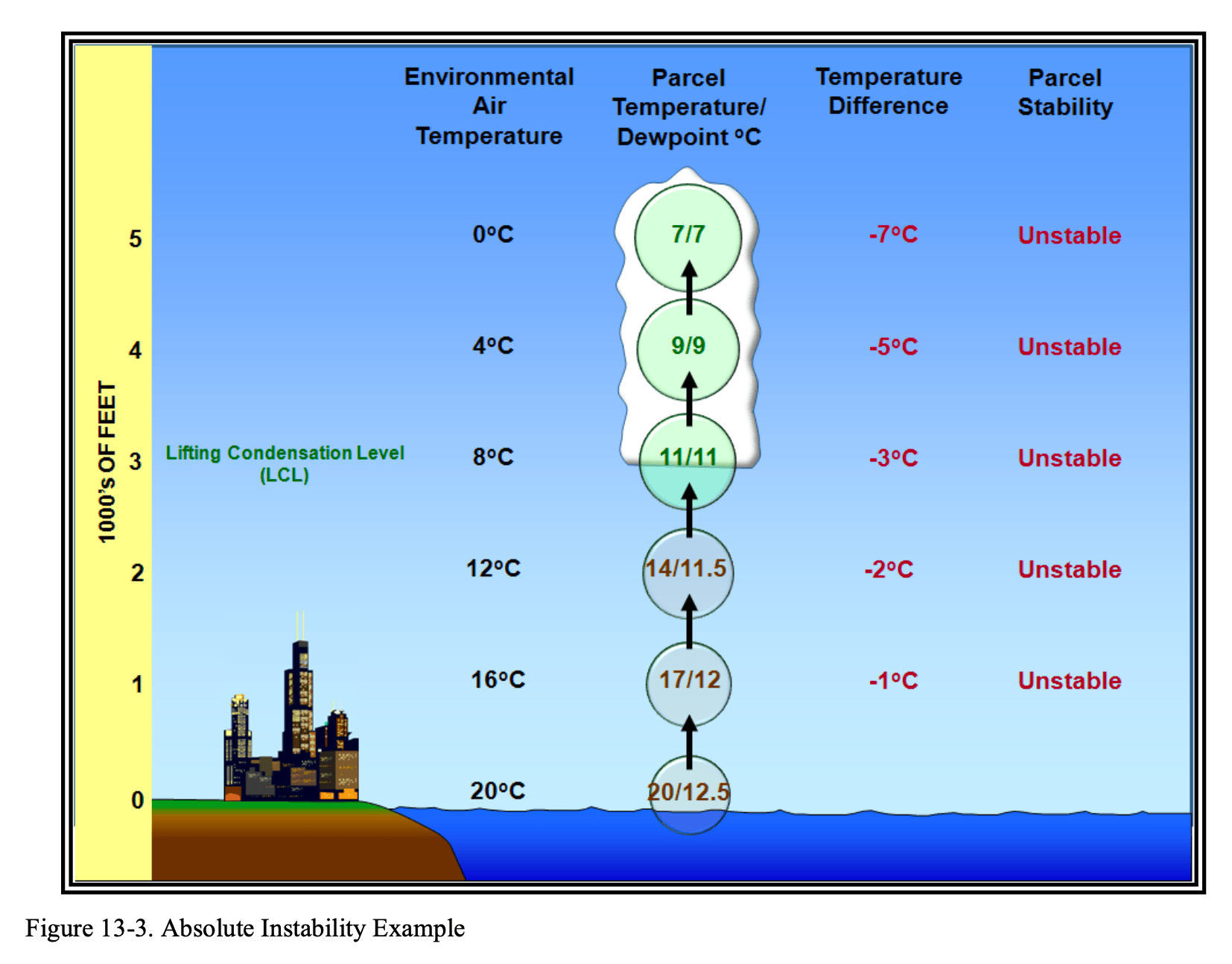
Absolute instability is, In this scenario, the surrounding environmental air is quite a bit cooler than the rising air parcel. Even while the parcel cools adiabatically, it remains warmer than the air around it. Because warm air wants to rise, this parcel will continue to ascend.
This often results in cumuliform clouds with “ruffled” edges.
Conditional Instability
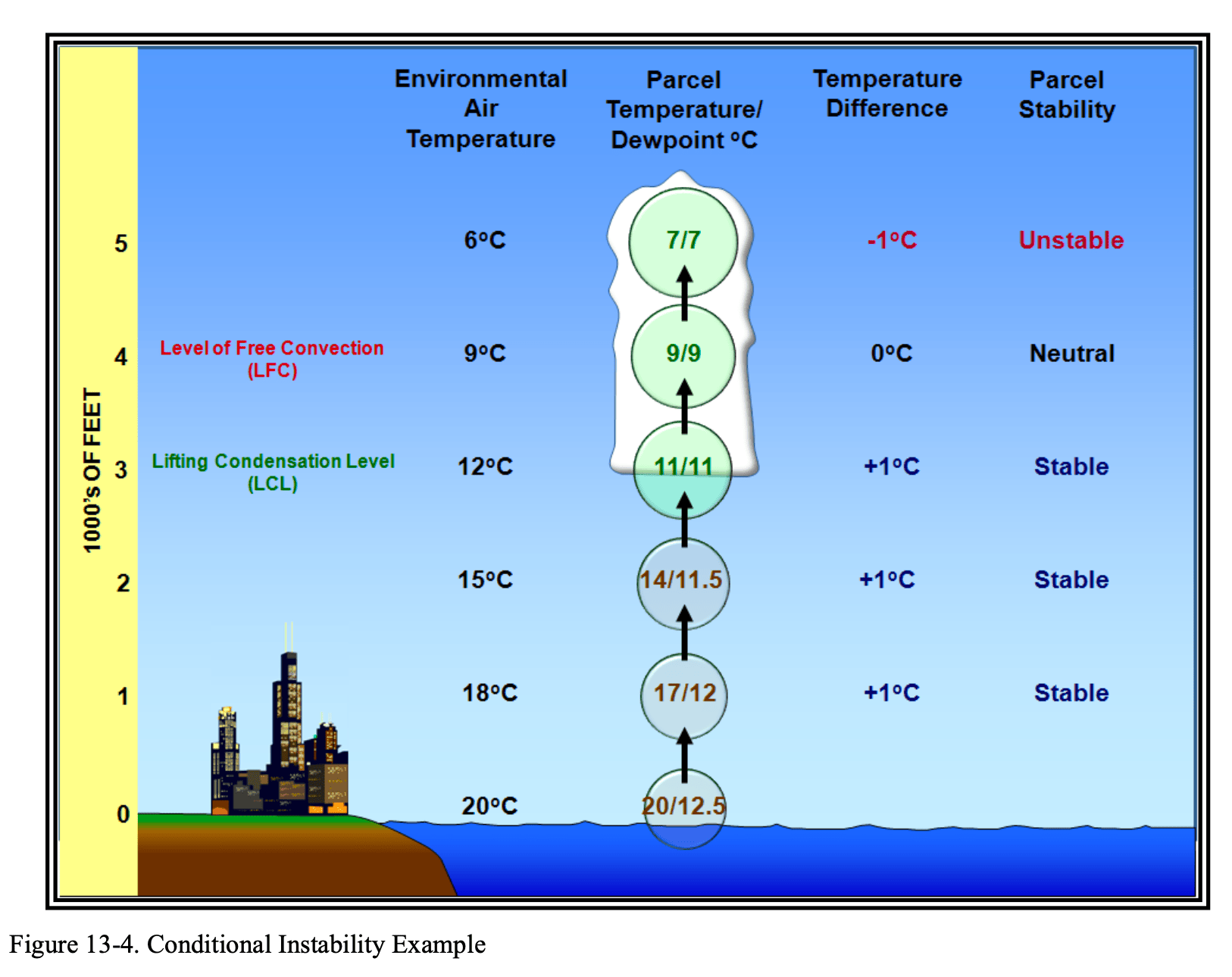
Conditional instability is the state of a column of unsaturated air in the atmosphere when its lapse rate of temperature is less than the dry adiabatic lapse rate but greater than the moist adiabatic lapse rate. An air parcel lifted upward would be initially stable, but at some point, above its LCL, it would become unstable. The term “conditional” means the parcel must be lifted to a particular level (altitude) before it becomes unstable and rises because of its own buoyancy.
The Level of Free Convection (LFC) is the level at which a parcel of air lifted dry adiabatically until saturated and moist adiabatically thereafter
would first become warmer than the surrounding environmental air (i.e., unstable) in a conditionally unstable column of air in the atmosphere. The LFC is a defining feature of a conditionally unstable column of air.
Summary of Air Stability
Simply stated, stability is how much resistance a layer of air will have rising vertically.
Stable air decreases the ability for it to move vertically resulting in stratus clouds and steady rain or snow. Unstable air increases the ability for air to move vertically resulting in cumulus clouds and showers developing. (I know it’s quite counterintuitive)
- Stable Air: - Stratiform clouds,
- possibly fog,
- continuous precipitation,
- smooth air (less turbulence),
- fair to poor visibility in haze.
- Unstable Air:
- Cumuliform clouds,
- showery precipitation,
- rough air (turbulence),
- typically good visibility (except in blowing obstructions).
Latent Heat
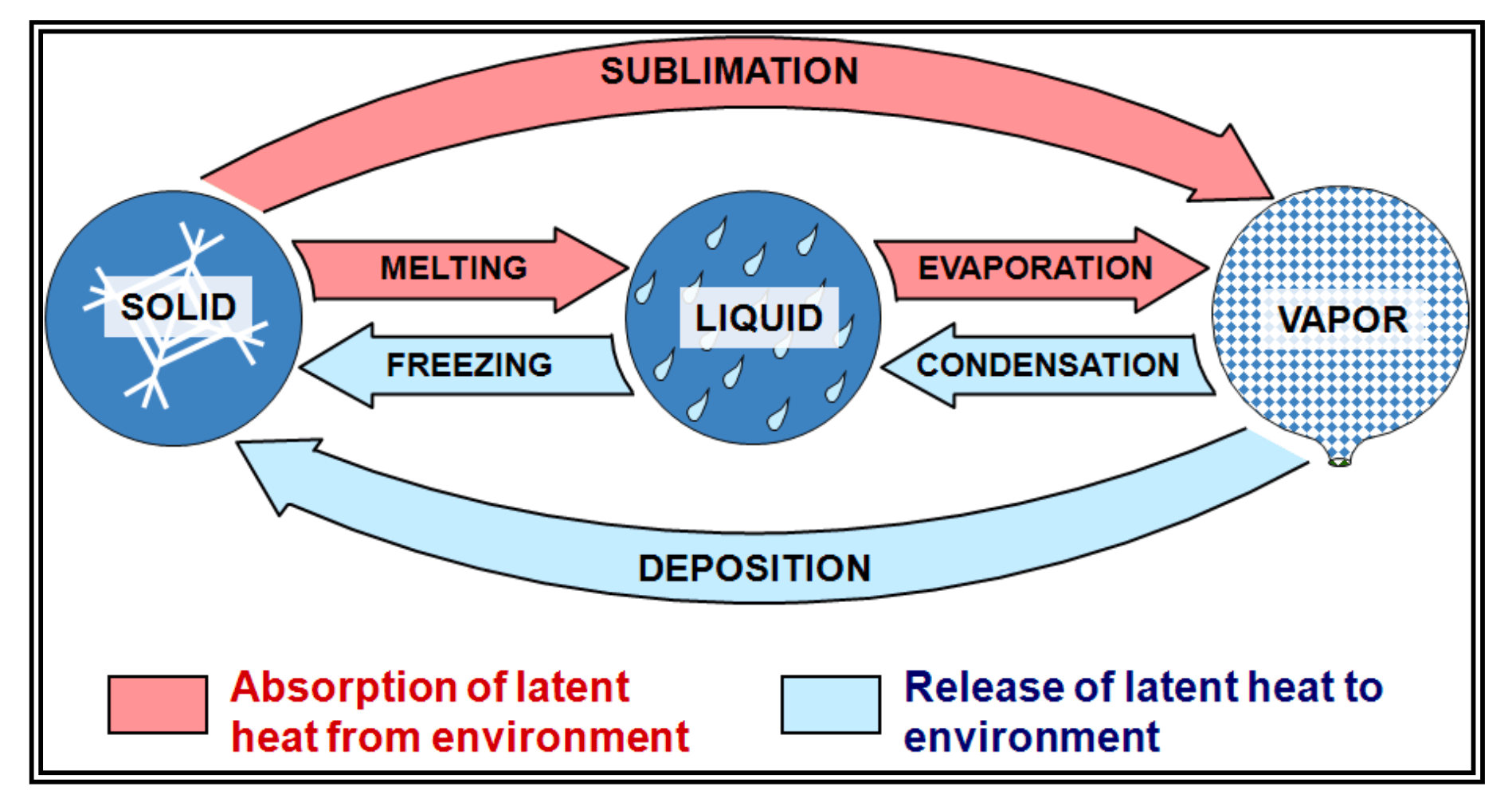
Latent heat is defined as the quantity of heat energy that is either released or absorbed by a substance when it undergoes a phase transition (changing state).
Energy Absorption (Cooling the Environment)
In these processes, water takes heat from the surrounding air to change its state. This results in a cooling effect on the air around the water.
- Melting: Solid (Ice) → Liquid
- Evaporation: Liquid → Vapor (Gas)
- Sublimation: Solid → Vapor (Gas)
Energy Release (Warming the Environment)
In these processes, water gives up its internal heat to the surrounding air. This results in a warming effect on the air around the water.
- Condensation: Vapor (Gas) → Liquid
- Freezing: Liquid → Solid (Ice)
- Deposition: Vapor (Gas) → Solid
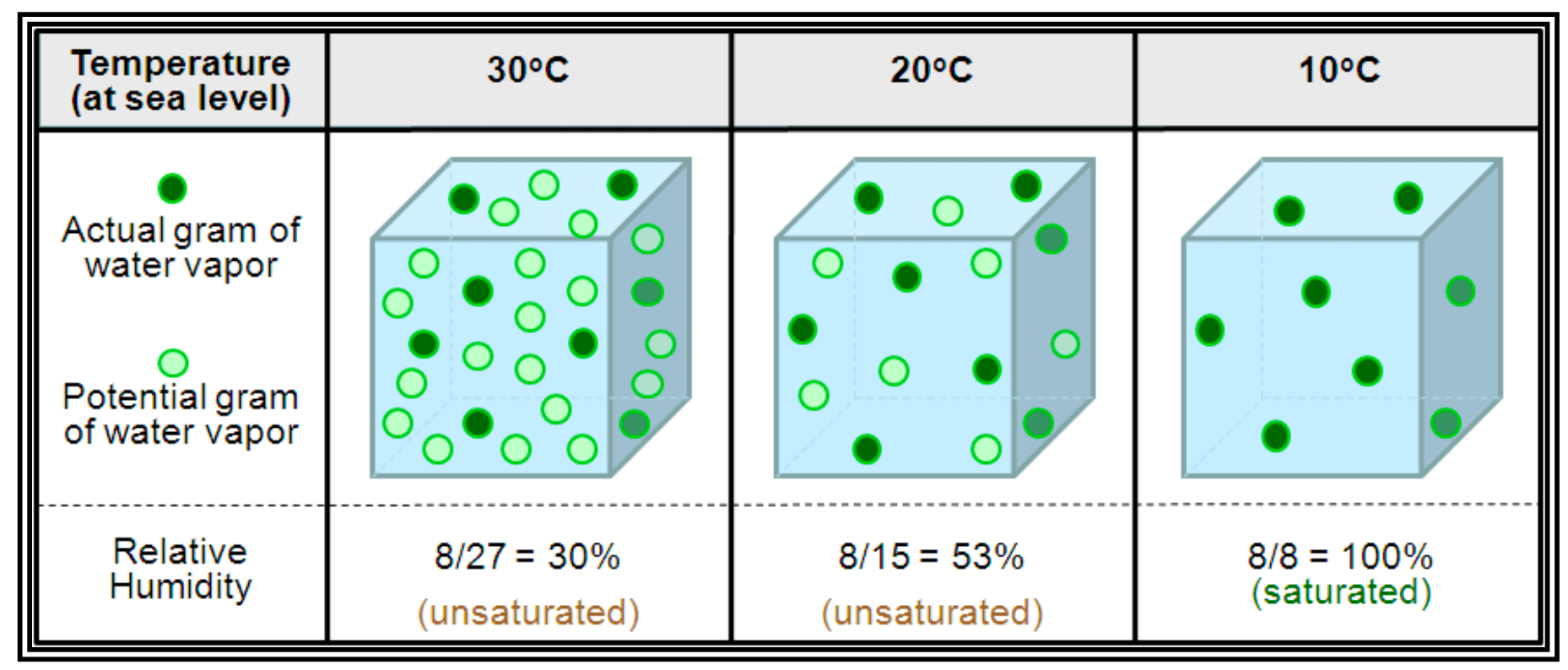
Humidity and Dew Point
As water evaporates, it changes from a liquid to a gas. The amount of water vapor in the air is humidity. It is important to remember that warm air can hold more water vapor than cold air.
- Dew Point: The temperature at which the air can hold no more moisture. When air is reduced to its dew point, it is saturated, and moisture comes out as fog, dew, or frost.
- Relative Humidity: The amount of water vapor actually in the air, expressed as a percentage of the total amount the air could hold at that specific temperature.
When the temperature and dew point are equal, the air is 100% saturated, and the relative humidity is 100%.
Keynotes:
- Adiabatic Process: A process where the temperature of an air parcel changes due to expansion or compression, with no heat exchange with the surrounding air.
- Stability: The resistance of the atmosphere to vertical motion.
- Instability: A state where air continues to rise because it is warmer (and less dense) than the surrounding environment.
- Convection: The vertical movement of air, specifically warm air rising.
- Latent Heat: Energy stored or released during a change in the physical state of water (e.g., liquid to gas).
- Sublimation: The transition of a substance directly from a solid state to a gaseous state.
- Deposition: The transition of a substance directly from a gaseous state to a solid state (the opposite of sublimation).
- Dew Point: The specific temperature to which air must be cooled to become saturated.
- Relative Humidity: The ratio of the actual amount of water vapor in the air to the maximum amount the air could hold at that temperature.
- Saturation: The point at which air holds the maximum amount of water vapor possible at its current temperature and pressure (100% relative humidity).
- Difference between latent heat and adiabatic heating? Latent heat is the energy absorbed or given up during a change of state (phase transition). Adiabatic heating is the heating resulting from a volume of air being compressed where no heat is exchanged with the surrounding environment.